Background
Allogeneic stem cell transplantation (allo-SCT) is widely regarded as the most effective consolidation treatment for patients with intermediate to high-risk ELN AML. However, despite its potential curative benefits, many elderly patients (aged ≥60) are unable to undergo this procedure due to comorbidities and/or inability to tolerate intensive chemotherapy and achieve a complete remission (CR). As a result, only a small proportion of these patients are able to benefit from allo-SCT.
Aims
The VEN-DEC GITMO trial is a prospective, multicenter, single arm, phase II study designed to assess the safety and efficacy of a “chemo-free” combination of Venetoclax and Decitabine (VEN-DEC) as a bridge to allo-SCT in elderly patients (60-75y) with intermediate to high-risk ELN AML. The primary endpoint of the study was the percentage of patients who underwent transplant in first CR. The study aimed to enroll 100 patients, with a projected dropout rate of 12%. The study was considered successful if 14 or more patients underwent transplant in CR.
Methods
Venetoclax (up to 400mg oral daily) plus decitabine (20 mg/m2 for 5 days every 28 days) were administered for 2 cycles. Patients who achieved complete remission (CR), defined according to ELN criteria as CR/CRi/MLFS, were required to undergo allo-SCT within 2 months. Patients who did not respond (NR) or achieved partial remission (PR) after C2 could receive an additional 2 cycles of the therapy, in order to achieve a CR prior to transplant. Patients who did not respond or achieved only a PR were discontinued from the study and could be treated according to the policies of their respective Centers.
Results
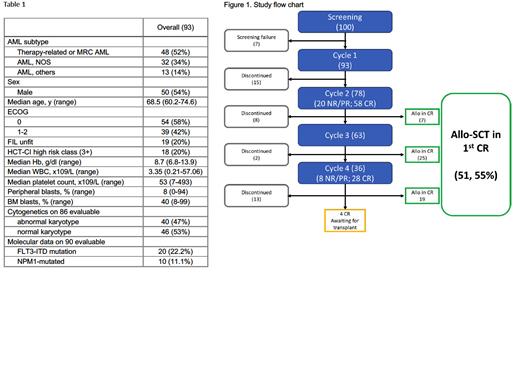
Overall, 100 patients were enrolled by 25 Italian GITMO transplant Centers as of December 2022. The data cut-off was on July 17th, 2023. Table 1 provides details on the cohort's characteristics. Notably, more than 50% of patients presented with myelodysplasia-related changes and/or therapy-related AML, and 47% of cases had an abnormal karyotype.
Seven patients (7%) were excluded due to screening failure. The remaining 93 patients started the 1 st cycle of treatment, and 78 patients completed the 2 nd cycle of VEN-DEC and were evaluable for treatment response.
Overall, 58 patients achieved complete remission (CR/CRi/MLFS) after 2 cycles (74%), while partial or non-response occurred in 20 cases. Four out of 20 (20%) PR/NR patients at C2 achieved CR after two additional cycles of therapy for a total of 62/93 (67%). Overall 51 and 26 patients received a third and/or a fourth cycle of treatment after achieving CR after cycle 2 while waiting for transplant, due to logistical issues. According to the study protocol, the 1 st cycle should be administered inpatient in all patients. C2, C3, and C4 were given outpatient in 79%, 93%, and 85% of cases, respectively. The median time to response was 65 days (range, 49-175). Overall, 38 (41%) patients were discontinued before transplant (7 cases for complications while on CR). A total of 19/93 (20%) patients died before transplant.
At the data cut-off, 51/93 (55%) treated patients and 51/62 CRs (82%) had successfully undergone allo-SCT. Figure 1 illustrates the course of study treatment.
A total of 139 grade ≥2 adverse events were reported in 56 patients (60%). Seventy events out of 139 (50%) were considered at least possibly related to study intervention. The following hematological toxicities were observed: prolonged grade ≥2 neutropenia was recorded in 34 out of 139 cases (24%), while anemia and decreased platelet counts were recorded in 7 cases (5%) and 3 cases (2%), respectively. Febrile neutropenia and infections were observed in 22 (16%) and 31 (22%) cases, respectively.
Conclusions
The results of the VEN-DEC GITMO trial demonstrate that the chemo-free VEN-DEC induction/consolidation regimen was highly effective, achieving a high rate of complete remission (CR 67%). Additionally, the regimen improved the feasibility of allo-SCT, with 55% of treated patients and 82% of those who achieved CR undergoing allo-transplantation (primary endpoint). Starting from C2, the regimen was administered in an outpatient setting in 80-90% of cases, which allowed patients to maintain their fitness and bridge to transplant in CR. The regimen also had a low clinical toxicity profile, with only 7 patients achieving CR discontinuing therapy due to adverse events.
Acknowledgement
Monica Bonzi, Sr. Clinical Lead - IQVIA RDS, Italy.
Disclosures
Russo:Medac, Abbvie, MSD, Jazz Pharma, Gilead, Novartis: Membership on an entity's Board of Directors or advisory committees; MSD, Novartis, Gilead, BMS, Medac: Honoraria. Polverelli:GSK: Honoraria; BMS: Honoraria; Abbvie: Honoraria; Novartis: Honoraria. Curti:Novartis: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Abbvie: Membership on an entity's Board of Directors or advisory committees. Vetro:BMS: Honoraria; Jazz Pharmaceuticals: Honoraria; ABBVIE: Honoraria. Borlenghi:Amgen, Incyte: Other: travel grants; AbbVie, BMS: Consultancy. Malagola:Biotest, MSD: Consultancy, Honoraria. Ciceri:ExCellThera: Other: Scientific Advisory Board .

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal